The carbon cycle or CO2 cycle is an important part of everyday life. Due to the abundance of carbon found in all elements of life, including animals, rocks, air, water, and more, the cycle of carbon is one that is constantly moving and changing due to the ever-changing nature of the things which carbon comprises.
Carbon is the fourth most common element found on the Earth and is responsible for the majority of human fuels and is found in many different reservoirs. The majority is found in the lithosphere, which is comprised of rocks. The majority of carbon is stored in sedimentary carbonates and kerogens, with the rest with the rest being spread between the ocean, the atmosphere, biomass such as plants and animals, and fossil fuels. This is known as carbon storage.
You will die but the carbon will not; its career does not end with you. It will return to the soil, and there a plant may take it up again in time, sending it once more on a cycle of plant and animal life.
– Jacob Bronowski
How Carbon Cycle Works?
Man-made activities like combustion of fossil fuels and heating of homes release carbon dioxide in the atmosphere. Respiration by humans and animals also introduce carbon into the atmosphere in the form of carbon dioxide. Plants have a process called photosynthesis that enables them to take this carbon dioxide from the atmosphere and water from the soil to form photosynthates (sugars, starches, carbohydrates, and proteins). Thus, carbon becomes the part of the plants.
Animals do not create their own food and eat plants or other animals to survive. The food they eat is broken down into sugars and starches by a process called metabolism. When the animals breather, the carbon dioxide is returned back to the atmosphere where plants use it again.
When both plants and animals die, they turn into fossil fuels which are made up of carbon over millions of years. This happens when decomposers like bacteria and fungi break down on to the dead plants and animals and release the carbon stored inside them. When we burn these fossil fuels, they come back to the atmosphere as carbon dioxide. Again plants will use carbon from the atmosphere and the whole process will start again.
Carbon dioxide is a greenhouse gas and traps the heat in the atmosphere. Without it and other greenhouse gases, this world will be a frozen planet. In the last 100 years, we have burned so much of fossil fuels that there is about 30% more carbon dioxide in the atmosphere today.
Carbon moves about from one storage type to another through multiple pathways of exchange which is why the burning of fossil fuels contributes to global warming: carbon released from fuels kept under the ground becomes infused with the air in the atmosphere, leading to a warmer climate.. The most important pathways upon which carbon travels are the atmosphere, the ocean, sediments and fossil fuels, the biosphere and the breakdown of biomass, and the interior of the earth.
Carbon exchanges between several different reservoirs of storage through multitudinous biological, geological, chemical, and physical processes. These processes are known as the carbon cycle.
The carbon cycle is split into two main types of movement, known as fast and slow cycles. Here is a breakdown of how the two work.
The Slow Carbon Cycle
The slow carbon cycle is the part of the cycle that sees carbon moving between rocks, the oceans, and soil. The process can take hundreds of millions of years, hence the name ‘the slow carbon cycle.’
- In order for carbon to move from the atmosphere to rocks, carbon is transmitted through rainfall. When the carbon mixes with the rainwater in the atmosphere, it creates a rain that is slightly acidic, due to the fact that the blend of carbon and other elements forms carbonic acid. This acid slowly dissolves rocks over a long period of time in a process that is more commonly known as ‘chemical weathering’. The rocks release chemical components such as magnesium, sodium, and calcium, which are then carried by rivers to the ocean.
- When these ions reach the ocean, they begin to combine with other elements to form new elements. Calcium, for example, fuses with bicarbonate ions which creates a new chemical called calcium carbonate. Calcium carbonate is a chalky white substance that is seen in cliff faces and in any area with hard water.
- Calcium carbonate is also created by creatures such as plankton and corals. When these organisms die, their carcasses fall to the floor of the ocean, where they become compressed over long periods of time by shells and sedimentary rock. This traps the carbon in newly generated rock such as limestone.
- On land, organic carbon is stored in mud. Great amounts of heat and pressure crush the mud down and compress the organic matter trapped within. After millions of years, these organisms and the mud in which they are encased will form rocks like shale, a type of sedimentary rock.
- Occasionally, with the right conditions, dead plant and animal matters can build up much faster than the existing layers can decay. When these conditions are met, it can often lead to the formation of other types of fuel storage such as oil, gas, or coal.
- In order for the carbon to return to the atmosphere whence it came, these rocks and fuels must be released through volcanoes. The rock and stone of the sea floor are constantly moving, and when they collide with one another, the immense heat and pressure can cause the rock to melt. When the rock is heated in this manner, it releases carbon dioxide.
- Volcanoes then erupt and release this carbon dioxide gas back into the atmosphere while also releasing swathes of silicate rock over the land. With the release of this new rock, the slow carbon cycle begins anew, with the carbon released into the atmosphere falling upon the new rock to return to the ocean and form new carbonates.
- The quickest element of the slow carbon cycle is the ocean, which actually vents out large amounts of carbon dioxide gas into the atmosphere, while also dissolving carbon dioxide gas it receives from the atmosphere. This steady exchange causes the ocean to produce hydrogen, thereby making it more acidic so that the water may weather the rocks found at the shores.
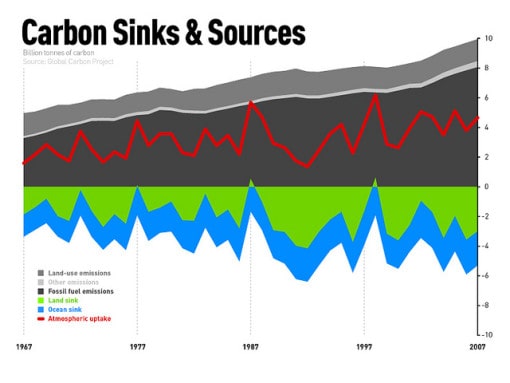
The Fast Carbon Cycle
Carbon is an essential element of all living things on Earth, and the exchange of carbons through these living organisms is what is known as the fast carbon cycle. While elements within the slow carbon cycle take hundreds of millions of years, the movements within the fast carbon cycle happen within the lifetime of a particular organism.
Within living creatures, carbon is one of the most important elements due to the number of chemical bonds in can form within each atom: up to four to each atom. This means that it can be combined with other chemicals in many complex ways. It is one of the most important parts of DNA, which comprises intertwined molecules anchored on a string of carbon atoms.
- The major organisms associated with the fast carbon cycle are plants and small ocean creatures known as phytoplankton. Both of these organisms take in the carbon dioxide found in the atmosphere and photosynthesize it to mix the carbon dioxide with water to create sugar and oxygen.
- There are four primary ways that the carbon found in plants can be released back into the atmosphere:
- Plants break down the sugar that they produce in order to stimulate growth
- Animals eat the plants (or, in the case of the ocean, the plankton), and they break down the sugar that is produced by these organisms in order to fuel their own growth or respiration
- The plankton or plants die and are consumed by bacteria and other micro-organisms in a state of decay
- The plants succumb to fire.
- In all of these examples, the sugar generated by these organisms combines with oxygen to create energy, as well as releasing water and carbon dioxide.
- When one of the above circumstances is met, the carbon dioxide typically ends up returning to the atmosphere. The fast carbon cycle is an essential part of the growth of plants and the life of plankton, and these organisms are responsible for a lot of the movement of carbon on earth. In winter times, when plants die out (or indeed in colder regions where they do not grow so readily), there is an observable rise in the concentration of carbon dioxide in the atmosphere. However, come spring time, these concentrations drop once more.
How Is The Carbon Cycle Changing?
The carbon cycle has always fluctuated and changed in response to major events such as changes to the climate. For example, when ice ages occurred in the past, the carbon cycle slowed down noticeably. When this happened, the coolness increased the growth of phytoplankton populations which added to the cooling effect on the earth. When the ice ages came to an end, the levels of carbon in the atmosphere rose drastically in response.
However, with the introduction of human industry, the carbon cycle is quickly becoming disturbed and its natural changes are being accelerated. The clearing of plants and the burning of fossil fuels releases more carbon into the atmosphere that should typically be released slowly, while not leaving behind the plants to absorb it.





